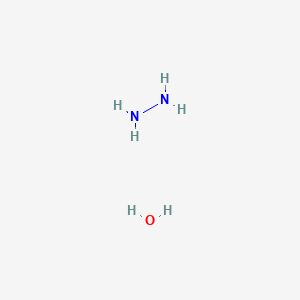
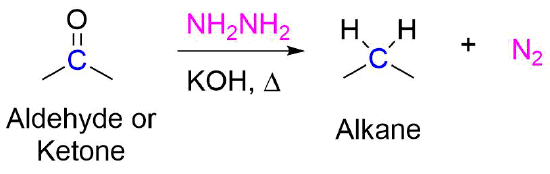Iron(III) Chloride‐Catalysed Aerobic Reduction of Olefins using Aqueous Hydrazine at Ambient Temperature - Lamani - 2012 - Advanced Synthesis & Catalysis - Wiley Online Library

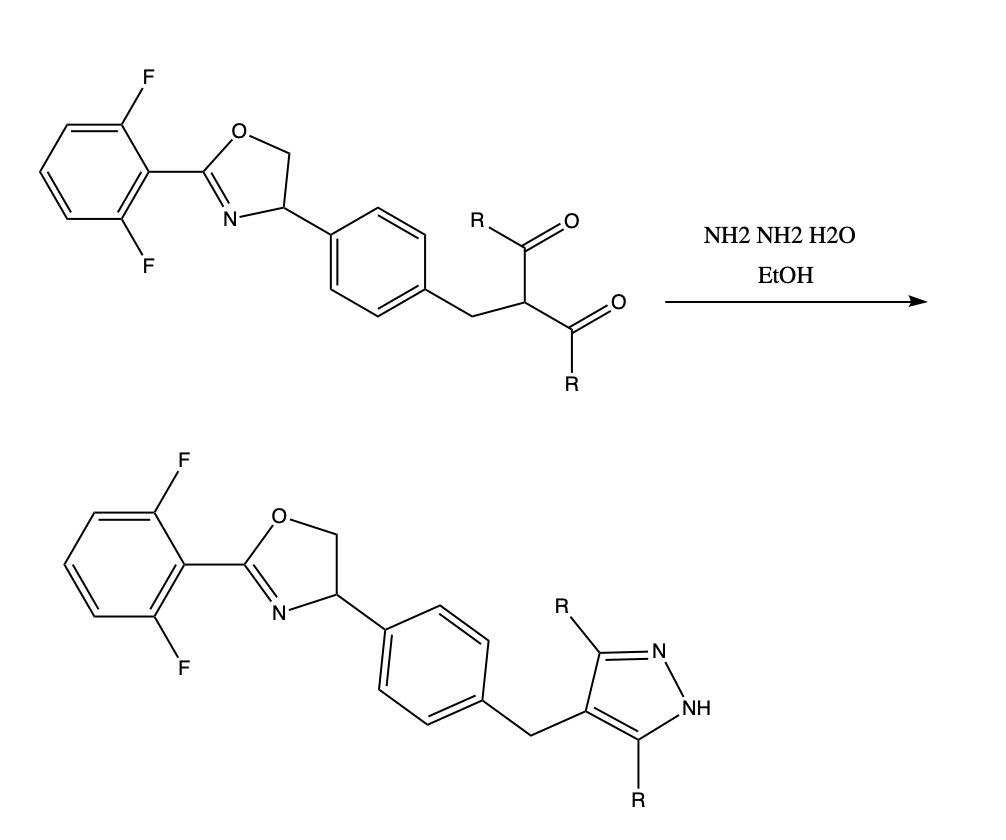
Functionalized 3-hydroxy-3-aminoquinoline-oxindole hybrids as promising dual-function anti-plasmodials - ScienceDirect
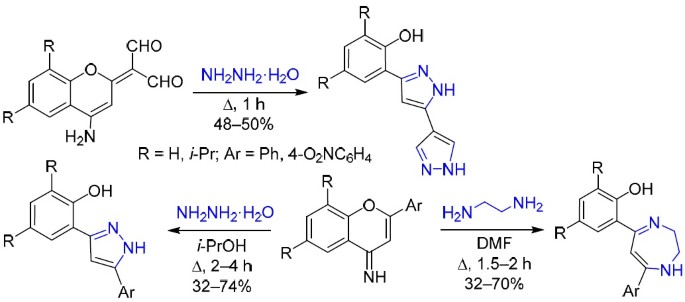
Reactions of 2Н(4Н)-chromenes with dinucleophiles: one-step synthesis of 2-(1H-(bi)pyrazol-3-yl)- and 2-(1,4(5)-(benzo)diazepin-4-yl)phenols | SpringerLink

Scheme 1 | Synthesis, Urease Inhibition, Antioxidant, Antibacterial, and Molecular Docking Studies of 1,3,4-Oxadiazole Derivatives

Conditions and reagents: (i) NH2NH2.H2O, EtOH, reflux, 6 h; (ii) DMF,... | Download Scientific Diagram

Identification of M‐NH2‐NH2 Intermediate and Rate Determining Step for Nitrogen Reduction with Bioinspired Sulfur‐Bonded FeW Catalyst - Zhao - 2021 - Angewandte Chemie International Edition - Wiley Online Library
![Rediscovering Bacon's hydrazine/phenylhydrazine mediated cyclization of 2,2′-dicarbonylbi(hetero)aryls: construction of (5-azo)-/indazolo[2,3- a ]quin ... - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D1OB02207E Rediscovering Bacon's hydrazine/phenylhydrazine mediated cyclization of 2,2′-dicarbonylbi(hetero)aryls: construction of (5-azo)-/indazolo[2,3- a ]quin ... - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D1OB02207E](https://pubs.rsc.org/image/article/2022/OB/d1ob02207e/d1ob02207e-s2_hi-res.gif)
Rediscovering Bacon's hydrazine/phenylhydrazine mediated cyclization of 2,2′-dicarbonylbi(hetero)aryls: construction of (5-azo)-/indazolo[2,3- a ]quin ... - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D1OB02207E
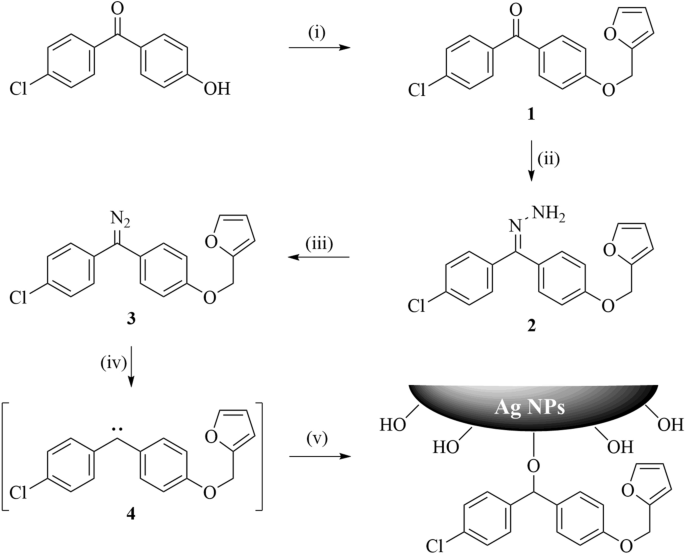
Carbene modification and reversible crosslinking of silver nanoparticles for controlled antibacterial activity | Scientific Reports
![Scheme 1, [(i) NH2NH2·H2O, EtOH (ii) PhC(O)CO2Et,...]. - Probe Reports from the NIH Molecular Libraries Program - NCBI Bookshelf Scheme 1, [(i) NH2NH2·H2O, EtOH (ii) PhC(O)CO2Et,...]. - Probe Reports from the NIH Molecular Libraries Program - NCBI Bookshelf](https://www.ncbi.nlm.nih.gov/books/NBK143197/bin/ml228f12.jpg)
Scheme 1, [(i) NH2NH2·H2O, EtOH (ii) PhC(O)CO2Et,...]. - Probe Reports from the NIH Molecular Libraries Program - NCBI Bookshelf
![5-Carbohydrazide and 5-carbonylazide of pyrazolo[3,4-b]pyridines as reactive intermediates in the synthesis of various heterocyclic derivatives - Ashraf A Aly, Talaat I El-Emary, Aboul-Fetouh E Mourad, Zainab Khallaf Alyan, Stefan Bräse, Martin Nieger, 5-Carbohydrazide and 5-carbonylazide of pyrazolo[3,4-b]pyridines as reactive intermediates in the synthesis of various heterocyclic derivatives - Ashraf A Aly, Talaat I El-Emary, Aboul-Fetouh E Mourad, Zainab Khallaf Alyan, Stefan Bräse, Martin Nieger,](https://journals.sagepub.com/cms/10.1177/1747519819861625/asset/images/large/10.1177_1747519819861625-fig2.jpeg)
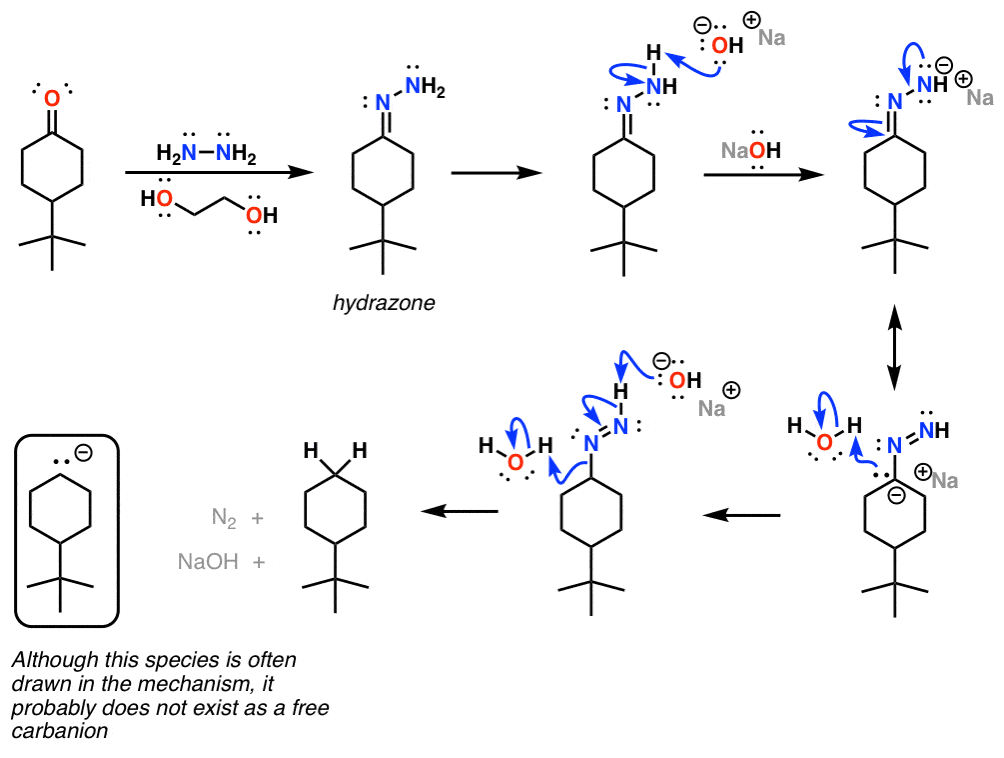
![Solved NH2NH2 H2O/HO- [heat] | Chegg.com Solved NH2NH2 H2O/HO- [heat] | Chegg.com](https://media.cheggcdn.com/media/2b8/2b86778d-b94b-43f3-ae4f-e5b21830cdfb/phpq7tc3G.png)