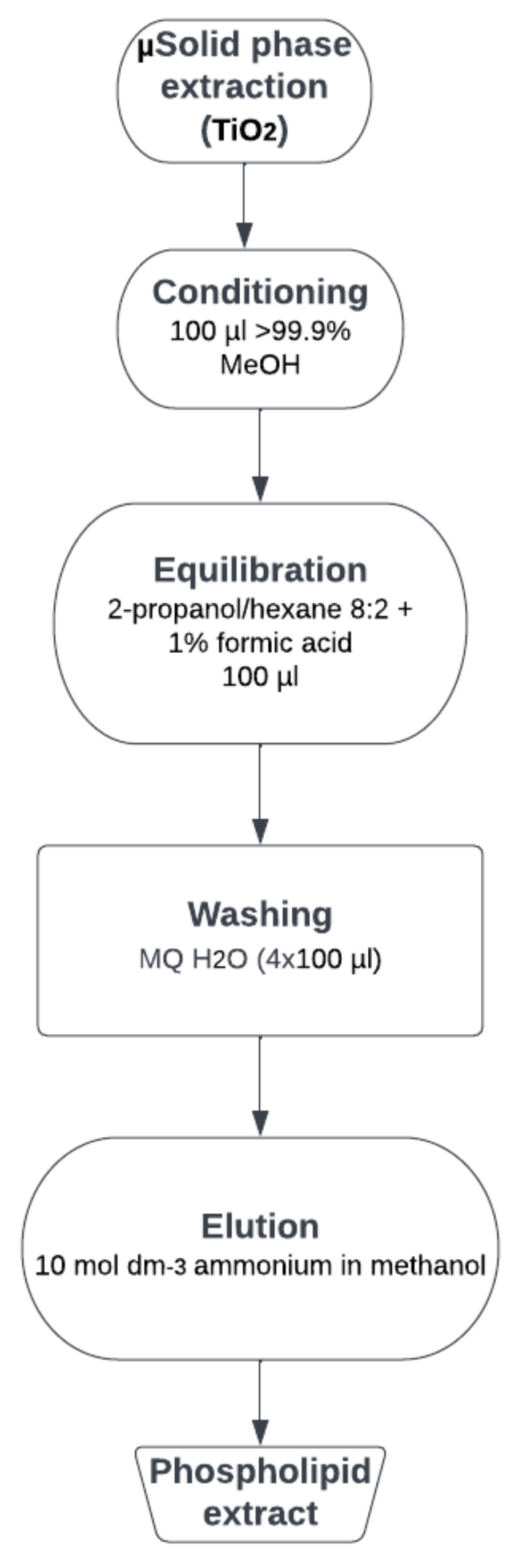
SOLVED: 4 Monoprotic base Q reacts in water by Q(aq) +HzO() = HQt(aq) + OH-(aq) (20 points). The pH of 0.073 M Q is 10.65. What is the value of Kb for

Mq Water Splash Waterdrop Waterdrops - Cleaning Water, HD Png Download - 1024x1024(#602035) - PngFind