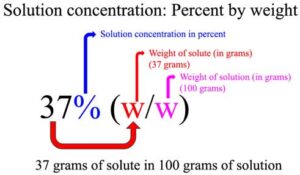
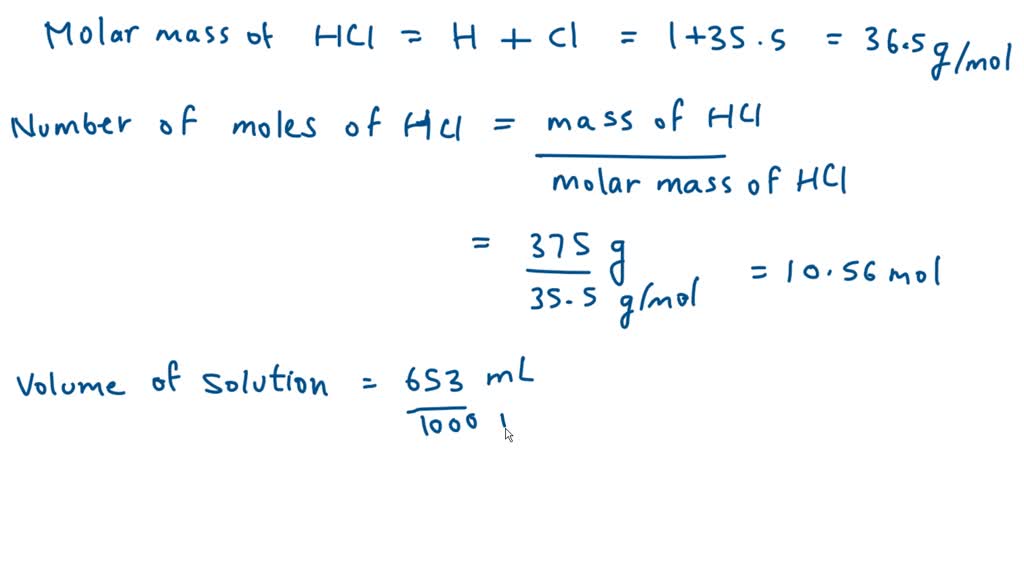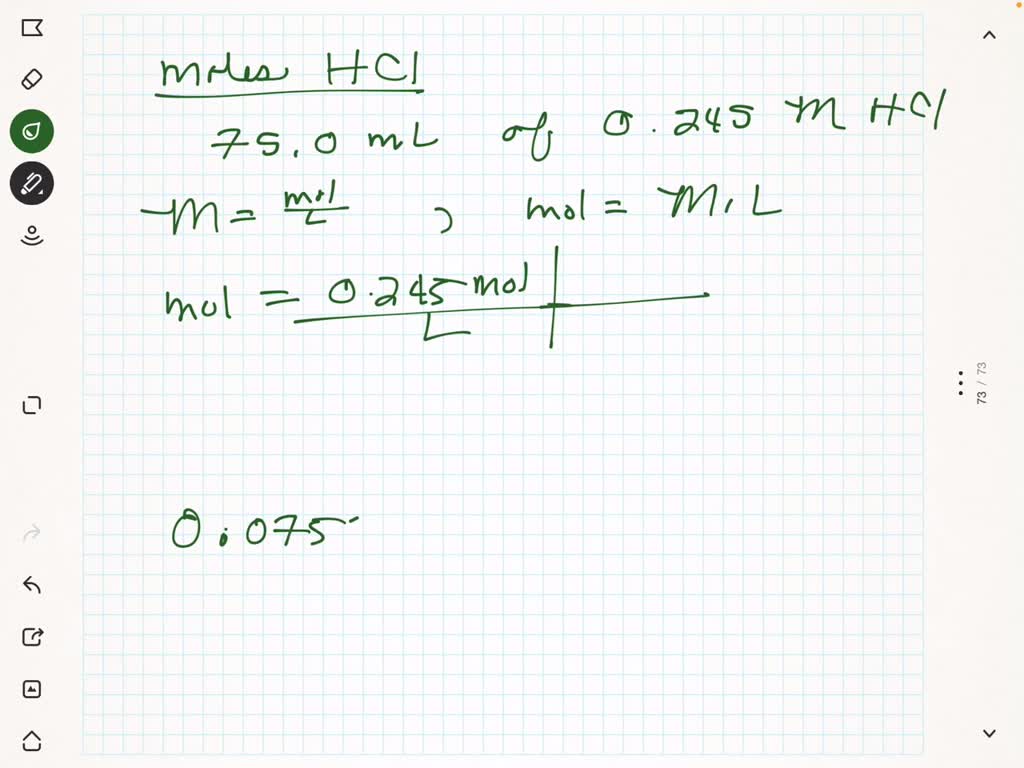SOLVED: Sample Exercise 13.6 Calculation of Mole Fraction and Molality An aqueous solution of hydrochloric acid contains 36% HCl by mass. (a) Calculate the mole fraction of HCl in the solution. (b)

SOLVED: Which has the most number of moles of HCl? a. 0.04L of a 0.1M HCl b. 0.01L of a 0.5M HCl c. 0.2L of a 0.2M HCl

Effect of HCl (0.1 to 2 mol/L) concentration on transport of As(III)... | Download Scientific Diagram

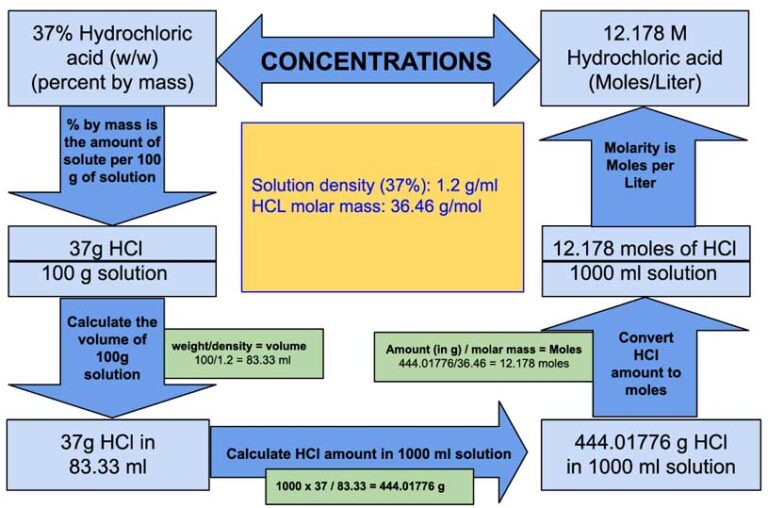
SOLVED: Q : Hydrochloric acid (FW=36.46 g/mol) is sold as 37% ww hcl with a density of 1.18 g/ml: The value of the solution 0.315mol of hcl is 26.3 ml 20.0 ml 2.0 ( 10.0 ( 2.6 ml ehogkky?

Hydrochloric Acid, c(HCl)=0.1 mol/L (0.1N) Titripur , MilliporeSigma, Quantity: 1L | Fisher Scientific

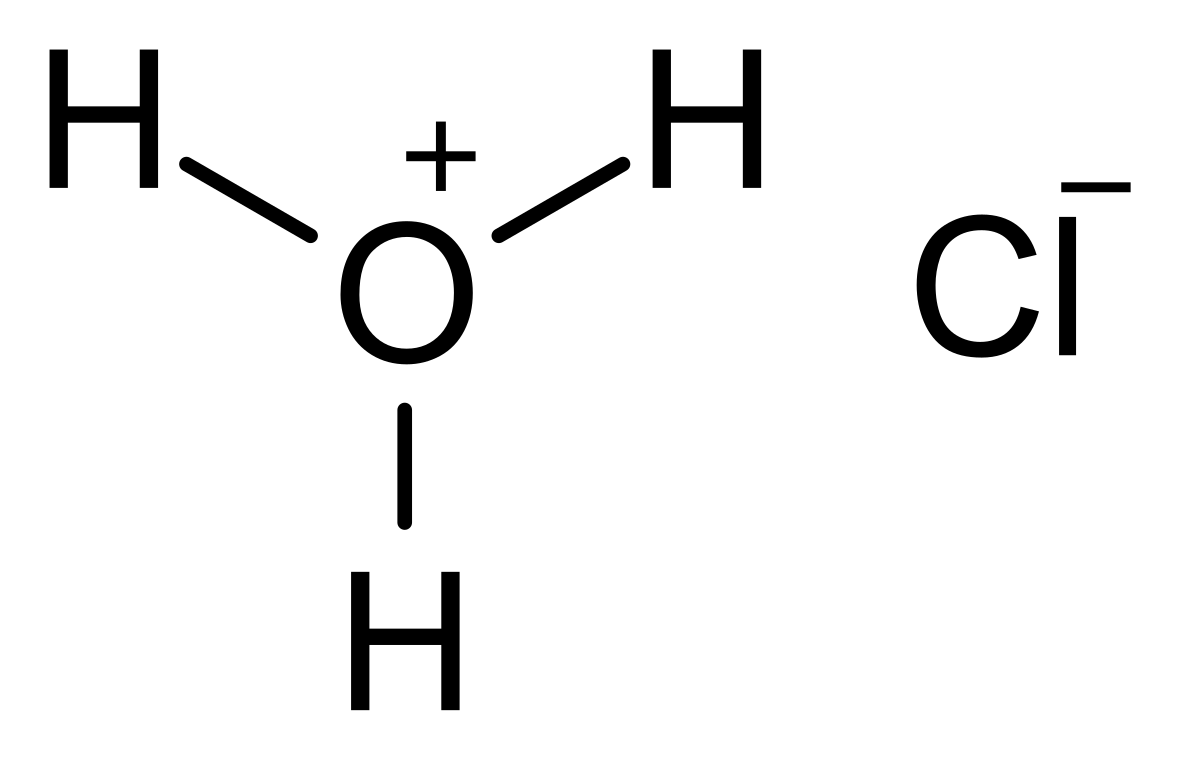
Lewis structure of HCl - How to draw Lewis structure of HCl, Polarity, Molecular Geometry and Hybridization along with FAQs