
Kobo Aura H2O Review: Weather Resistance And A Great Screen Combine For Ultimate E-Reading | TechCrunch
The standard reduction potential of the reaction at 25^° C ,Н2О + e^ gives half H2(g) +OH^ is 0.8277 V. The equilibrium constant for the reaction H2O reversible H3O^++OH^ at 25^° C is

Amazon.com: kwmobile Cover for Kobo Libra H2O - Fabric e-Reader Case with Built-in Hand Strap and Stand - Dark Grey : Electronics

H2O Related Processes Dissociative Recombination 1. H3O + + e − → H2O +... | Download Scientific Diagram

E-pH Diagrams for the Li-Fe-P-H2O System from 298 to 473 K: Thermodynamic Analysis and Application to the Wet Chemical Processes of the LiFePO4 Cathode Material | The Journal of Physical Chemistry C


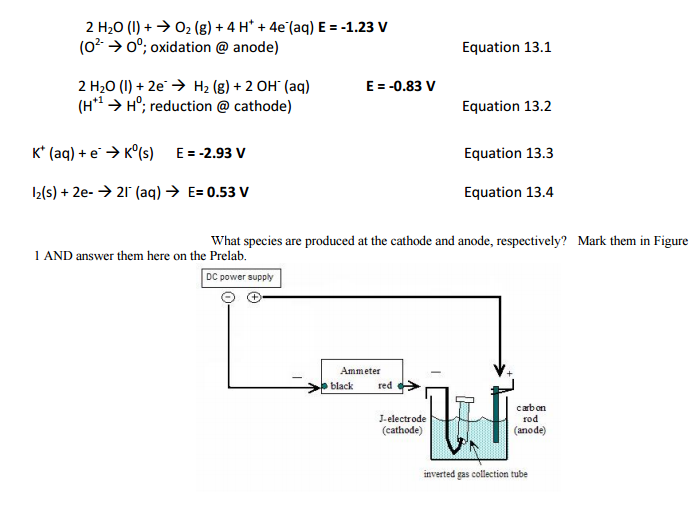

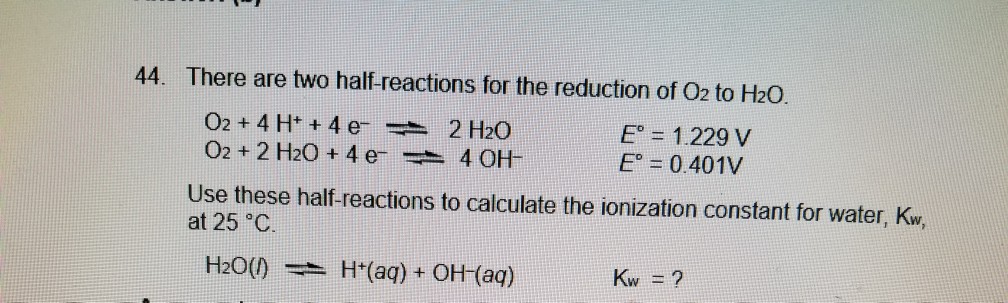



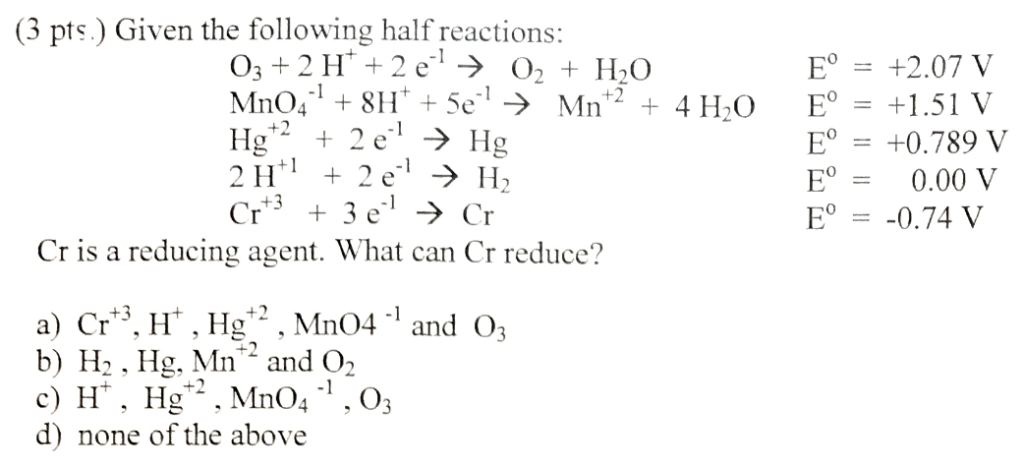



:max_bytes(150000):strip_icc()/Kobo_EReaders_1LW4046778_HeroSquare-ea9a5889b98f49fa8e64f46c7199f5b8.jpg)

