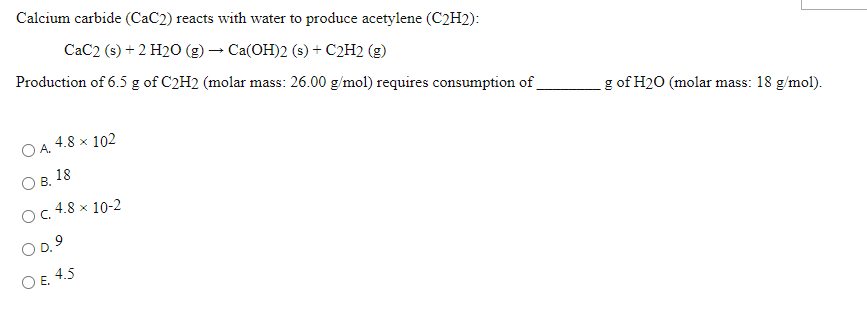Class11Cac2+H2O=acetylene (calcium carbide react with water)complete reaction explanations in Telugu - YouTube

In the given reactions, Be2C + H2O to BeO + X CaC2 + H2O to Ca(OH)2 + Y Mg2C3 + H2O to Mg(OH)2 + Z X, Y and Z are respectively.

polyethylene can be produced from CaC2 according to following sequence CaC2+ H2O-CaO + HCtriple bond CH n(HC triple bond CH) - Chemistry - Basic Concepts in Chemistry - 10693899 | Meritnation.com

Towards C1 chemistry: methanol vinylation by CaC2 in water in the presence of potassium or sodium carbonates - Parshina - 2019 - Journal of Chemical Technology & Biotechnology - Wiley Online Library

From the following reactions at 298 K .(A) CaC2(s) + 2H2O(l) → Ca(OH)2(s) + C2H2 (g); Δ H^∘ = - 127.9 kJ mol^-1 (B) Ca(s) + 12 O2(g) → CaO(s) ; Δ

Chemical Equations And balancing equations. Chemical Equation CH 4 + O 2 CO 2 + H 2 O Reactantsproducts Means to produce. - ppt download

CaC2 + H2O → CaO + HC≡ CH n(CH ≡ CH) + nH2 → - (CH2 - CH2)n - Polyethylene can be produced from calcium carbide according to the given sequence of
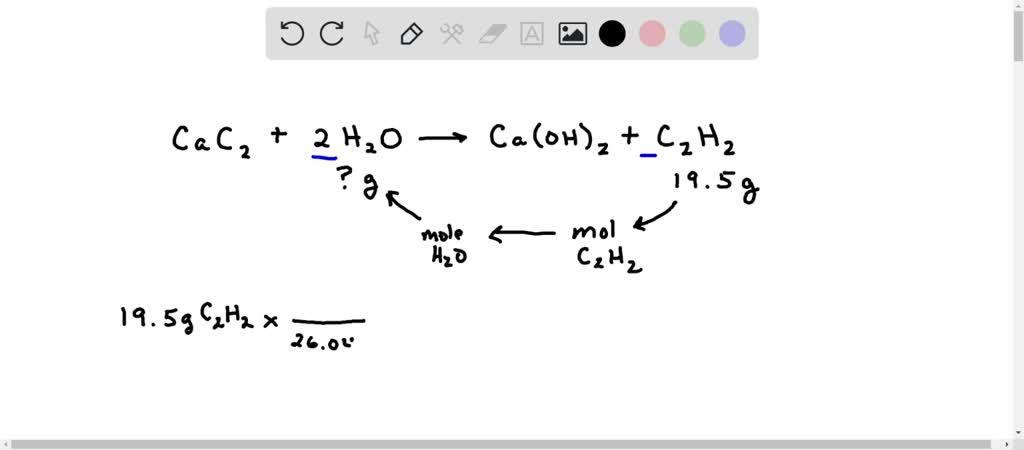
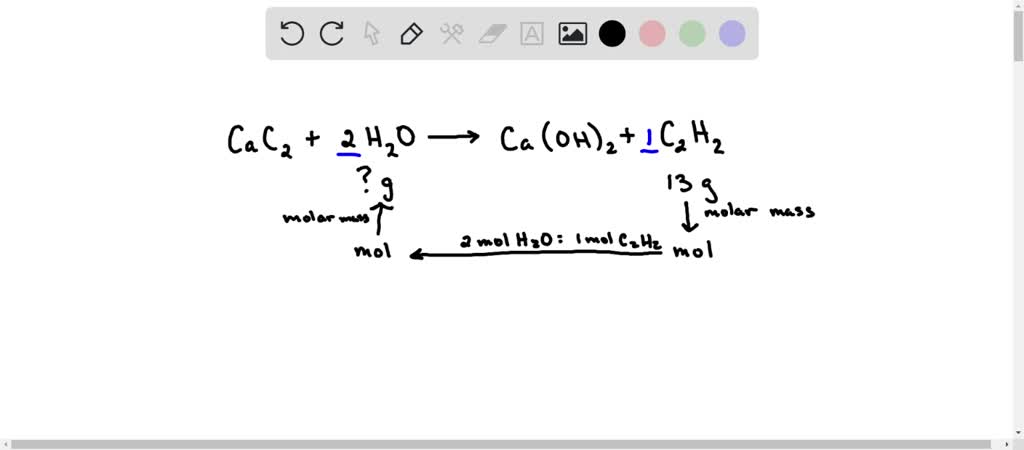
SOLVED: Calcium carbide (CaC2) reacts with water to form acetylene (C2H2): CaC2 (s) + 2 H2O (g) → Ca(OH)2 (s) + C2H2 (g) How many grams of water are required to produce